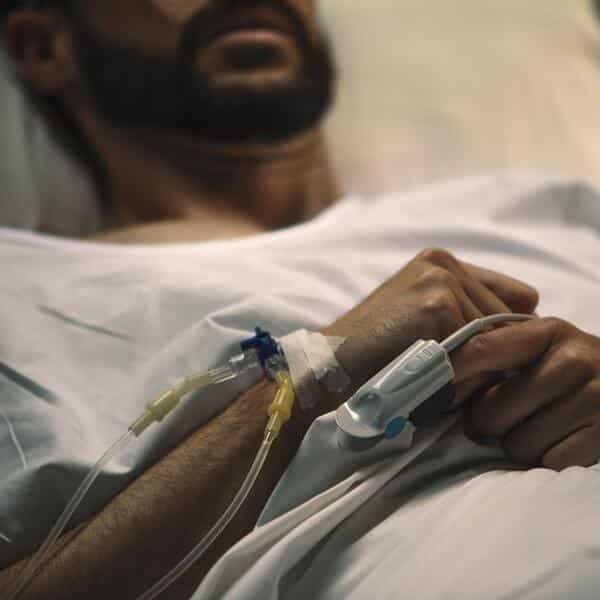
At the Stem Cells Transplant Institute in Costa Rica, we offer stem cell therapy programs for critical limb-threatening ischemia (CLTI) and peripheral artery disease (PAD) designed to support circulation, vascular health, and tissue viability through regenerative medicine approaches. MSC-based protocols may help support inflammatory regulation and activate the body’s natural tissue repair processes when used alongside conventional vascular care.
Therapeutic Angiogenesis Using Bone Marrow Cell Transplantation
Randomized controlled trial evaluating autologous bone marrow cell transplantation for limb ischemia, demonstrating improved blood flow and clinical outcomes.
 View published study
View published study